Debate intensifies over mass vaccination drives for prevention of cervical malignancy; experts emphasize scientific evidence while critics call for greater transparency in clinical evaluation.
Dainik india news,Lucknow.The expanding immunization initiatives targeting adolescent girls for the prevention of Cervical Cancer have increasingly become a subject of clinical debate, ethical scrutiny and public health discourse. While the biomedical community largely endorses the prophylactic role of the Human Papillomavirus vaccine, certain observers and commentators have begun raising questions regarding regulatory transparency, long-term immunological outcomes and the communication strategy surrounding large-scale vaccination campaigns.
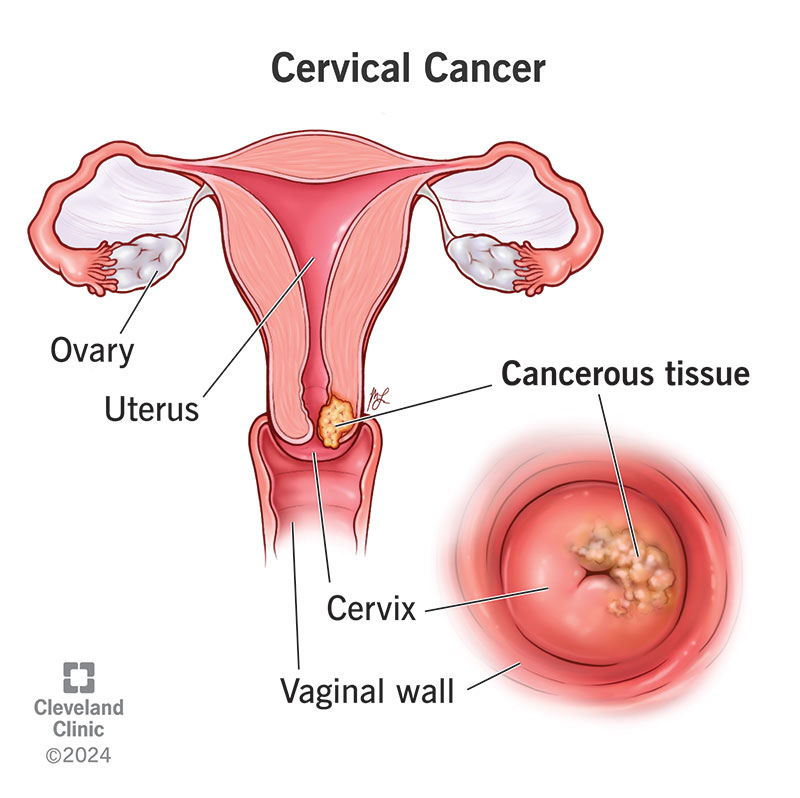
From a biomedical standpoint, cervical carcinoma is strongly associated with persistent infection by Human Papillomavirus, a group of oncogenic viral strains known to induce premalignant epithelial transformations within the cervical mucosa. Consequently, prophylactic immunization against HPV has been advocated by many global health agencies as a preventive strategy aimed at reducing the burden of cervical neoplasia, particularly when administered during early adolescence prior to viral exposure.
However, the expanding discourse surrounding the vaccine has also brought forth a series of critical inquiries regarding the nature and visibility of clinical trial protocols, pharmacovigilance mechanisms and post-marketing surveillance data. Critics argue that while vaccines undergo multi-phase clinical trials—including Phase I safety assessments, Phase II immunogenicity evaluation and Phase III randomized controlled trials—greater public accessibility to these datasets may help address lingering concerns among sections of the population.
In addition, some public health commentators emphasize that contemporary pharmaceutical innovation operates within a complex ecosystem that involves regulatory agencies, multinational pharmaceutical corporations and national immunization programs. In such an environment, they argue, the ethical imperative of maintaining transparent communication with the public becomes even more significant.
Another dimension of the debate concerns the communication strategy adopted during mass vaccination campaigns. Public health drives often involve intensive awareness programs, institutional endorsements and community outreach initiatives designed to maximize vaccine uptake. Critics contend that in certain circumstances the emphasis on rapid coverage may overshadow detailed public understanding of immunological mechanisms, potential adverse events and long-term monitoring protocols.
Medical professionals, however, strongly contest the notion that vaccines are introduced without adequate evaluation. According to immunologists and epidemiologists, modern vaccine development is governed by stringent regulatory frameworks that involve toxicological studies, immunogenicity analyses, randomized clinical trials and extensive safety monitoring before authorization for public use. Furthermore, global health organizations maintain that HPV vaccination has demonstrated significant efficacy in preventing high-risk viral infections responsible for cervical malignancies.
Experts also highlight the importance of pharmacovigilance systems, which continuously monitor vaccine safety through adverse event reporting networks and epidemiological surveillance. These mechanisms, they argue, ensure that any unexpected clinical patterns are rapidly investigated and addressed through scientific inquiry.
Nonetheless, the broader conversation surrounding adolescent immunization ultimately reflects a fundamental principle of public health governance: scientific progress must be accompanied by public trust. When citizens are provided with comprehensive information about clinical trials, immunological benefits, potential risks and regulatory safeguards, informed decision-making becomes possible.
Thus, the ongoing debate over HPV vaccination should not merely be viewed as a confrontation between scientific authority and social skepticism. Rather, it represents an opportunity to reinforce transparency, strengthen evidence-based communication and deepen public engagement in healthcare policy.
In the evolving landscape of preventive medicine, where biotechnology continues to reshape the boundaries of disease prevention, maintaining equilibrium between innovation, ethical responsibility and public confidence remains the central challenge for health systems worldwide
